New Battery of Chemical Battery: Peking University Develops Asymmetric K/Li Dual-Ion Battery
In recent years, with the rapid growth in the demand for power batteries, the price of raw materials in the upper reaches has also increased rapidly. In particular, raw materials such as Li, Co, and Ni have all experienced substantial price increases in 2017, which greatly compressed lithium-ion batteries. The profit margin has also become an impediment to the further decline in the cost of lithium-ion batteries. In order to reduce the production cost of lithium-ion batteries, new types of low-cost batteries including K-ion batteries and Na-ion batteries have been rapidly developed in recent years, and cathode materials and electrolytes have made great progress, but due to the ion radius of K+ and Na+. It is relatively large, so the choice of negative materials available for embedding is still a challenge, hindering the development and application of these low-cost batteries. In order to solve the problem of poor performance in the embedding of K, Na ions in traditional materials such as hard carbon, Jiaxin Zheng et al. of Beijing University, based on the traditional K-ion battery, optimized the battery's formula to achieve different types of positive and negative batteries. The reaction: The positive electrode is still subjected to the insertion and extraction of K+, but the negative electrode uses graphite material for the insertion and extraction of Li+. This not only takes into account the low cost of the K-ion battery, but also takes into account the high performance of the Li-ion battery.
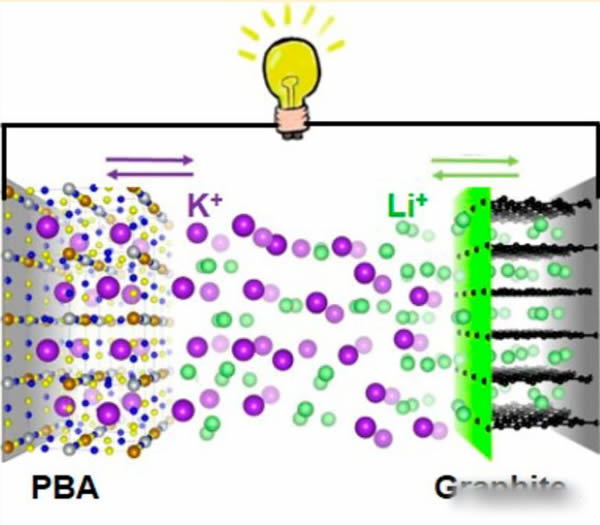
We know that there are three major parts of lithium-ion batteries that really participate in electrochemical reactions: positive electrodes, negative electrodes, and electrolytes. When a lithium-ion battery is charged, two reactions occur simultaneously: 1) The Li+ of the positive electrode escapes and enters the electrolyte. 2) Li+ in the electrolyte is embedded in the negative electrode. It should be noted that these two reactions happen at the same time, that is, Li+ in the electrolyte is embedded in the negative electrode under the driving of the electric field, and at the same time, the Li+ that comes out of the positive electrode will replenish the reduced Li+ in the electrolyte. We have provided new ideas for optimizing the structure of K-ion batteries. Jiaxin Zheng's proposal is that cathode materials for K-cells are still used in cathode materials, but the electrolyte needs to use a mixed electrolyte containing both K+ and Li+, and the cathode uses a traditional lithium-ion battery anode material—graphite material. In this way, during the charging process, K+ emerges from the positive electrode and enters into the electrolyte. Li+ in the electrolyte is embedded in the graphite (because the radius of the K+ ion is too large to be embedded in the graphite), such a design maintains K. While the ion battery has low cost, it also ensures good cycle performance of the battery, the battery operating voltage reaches 3.6V, and there is no significant decline in the cycle of 5000 times.
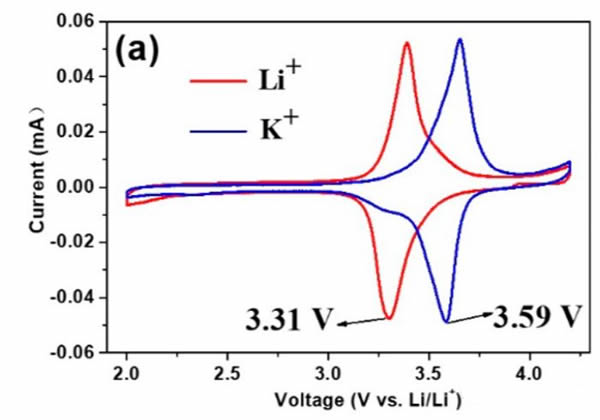
In the selection of positive electrode materials, Jiaxin Zheng chose the common K+ battery cathode material K2NiFeII(CN)6, which is different from the basic mechanism of interaction between alkali metal ions and O in cathode materials of Li-ion batteries. K-ion battery cathode materials (such as K2NiFeII) The alkali metal ions in (CN)6) will interact with 12 CNs through p electrons. Through computational studies, it has been shown that increasing the ionic radius of the alkali metal can increase the voltage plateau at which the alkali metal ions are embedded in the interaction with p-electrons, which is also verified in cyclic voltammetry tests (as shown in the figure above). The voltage at which K+ is embedded in the material (3.59V) is 0.28V higher than the voltage at which Li+ is embedded (3.31V), so K+ will be embedded in the positive electrode material first during discharge. However, because the negative electrode is affected by a short radius of K+, the negative electrode is almost only embedded in Li+, which ensures that Li+ is only embedded in the negative electrode and K+ is only embedded in the positive electrode. The positive and negative electrodes do not affect each other.
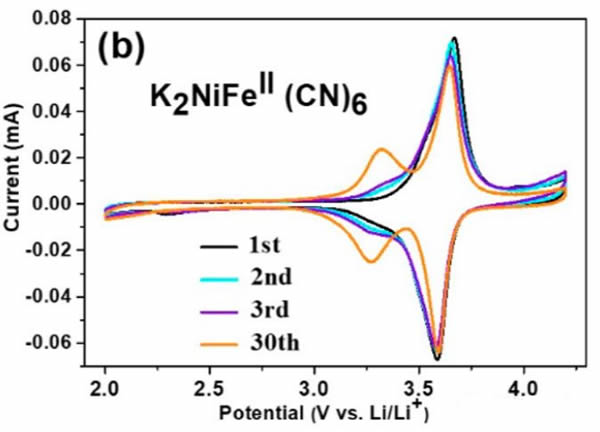
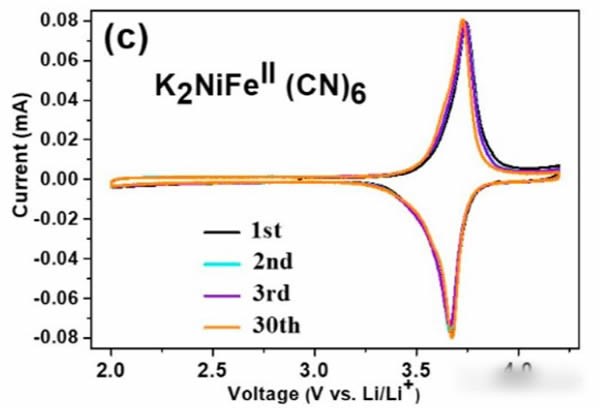
From the figure below, when using K2NiFeII(CN)6 as the positive electrode (LiPF6 with 1mol/L electrolyte), almost only the K+ embedded current peak near 3.59V can be seen in the cyclic voltammogram at the beginning. It shows that only K+ will be embedded in the cathode material at this time, but after cycling 30 times, there is also a current peak at the 3.3V cathode. This is mainly because the K+ concentration in the electrolyte is too low, resulting in partial Li+ embedding. Among the cathode materials. If a part of KPF6 is added to the electrolyte, Li+ can be effectively prevented from intercalating into the positive electrode material (as shown in Figure c, the electrolyte is 0.5 mol/L KPF6 and 0.5 mol/L LiPF6 mixed solute).
In terms of anode materials, Jiaxin Zheng chose a common cathode material for commercial lithium-ion batteries—graphite. According to previous research results, since the ion radius of Li+ is significantly smaller than that of K+, Li+ is more easily embedded into graphite materials, so when When the graphite material is used as a negative electrode of an asymmetric K/Li double-ion battery, it can be ensured that the negative electrode only inserts Li+, and the positive electrode only inserts K+.
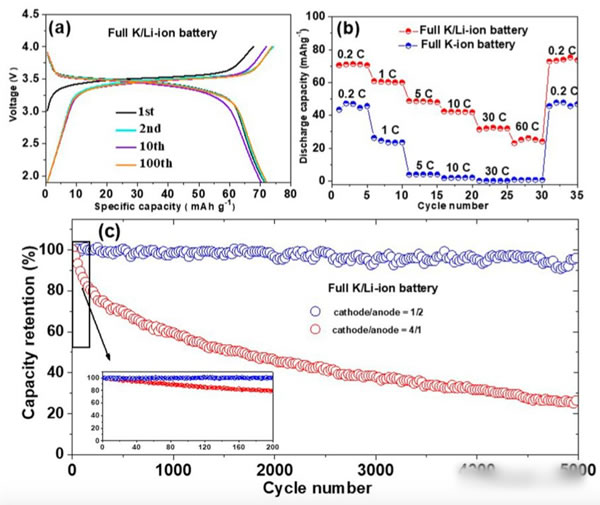
The following figure shows the electrochemical performance of the asymmetric K/Li dual-ion battery designed by Jiaxin Zheng. The positive electrode uses K2NiFeII(CN)6, and the negative electrode uses graphite (pre-lithiated to avoid SEI. Li) is consumed during membrane formation. The electrolyte is 0.5M KPF6 and 0.5M LiPF6.
As can be seen from Figure a, the battery voltage platform in the vicinity of 3.6V, the corresponding K + in the positive electrode of the embedding and the reaction, in the cycle after 100 times the positive electrode capacity still maintained at 71.2mAh / g, and the theoretical value of 70.7mAh /g is very close. Compared to pure K-ion batteries, the rate performance of asymmetric Li/K dual-ion batteries has been significantly improved (see Figure b below). The most critical is that the battery also showed excellent cycling performance, and there was no noticeable decline in the number of cycles of 5000 cycles at a magnification of 30C (as shown by the blue curve in Figure c below). At the same time, we also noticed that the mass ratio of positive and negative electrodes has a significant effect on the cycle performance of the battery. The increase in the number of negative electrodes (mass ratio: positive electrode: negative electrode = 1:2, and the capacity of the negative electrode is 8 times that of the positive electrode) can be significant. In order to improve the cycle performance of the battery, Xiaobian believes that the main problem that leads to excessive usage of the negative electrode is that the selectivity of the graphite to the two ions is not as good as one would think. Therefore, when the graphite negative electrode is too small, it will cause partial K+ insertion during the cycle. The decrease in cycle performance can be achieved by means of artificial selective SEI membranes to increase the selectivity of the negative electrode and reduce the amount of graphite negative electrode.
Jiaxin Zheng's work provides a new idea for the design of K-ion batteries. If we can't find a suitable anode material for K+ embedding, then we simply use the cathode of a Li-ion battery. The anode and the cathode work independently. Impact, effectively ensure the performance of the battery. Although this technology is not yet mature, there are still many problems to be solved. For example, the cathode material has a low capacity and the negative electrode capacity design requirements are relatively high. However, as the technology matures, these problems can be solved step by step. The idea can help us better promote the design of other such as sodium ion batteries, promote the popularization and application of new batteries.
Led Moving Head Wash,Zoom Moving Head Light,Bee Eye Moving Head Light,Zoom Moving Head Wash Light
EV LIGHT Guangzhou Co., Ltd , https://www.evlightpro.com
